MIFETM
System Overview
This page outlines the theory and has information
on the MIFE system, the CHART
and MIFEFLUX software.
Microelectrode ion flux measurement - principles and
basic theory
Chemicals in solution move under the influence of chemical forces of diffusion
directed towards lower concentration regions. Ions, which are charged,
also experience electrical forces if an electric field is present as well.
The movement of an ion in solution can be described in terms of these chemical
and electrical driving forces and other parameters of the ion and solution.
It can be shown that the net flux of an ion, typically measured in units
of nmol m-2 s-1, may be found from a measurement
of the change in voltage of an ion selective microelectrode that is moved
through a small known distance in the solution. This technique allows non-invasive
measurement of net ion fluxes through a tissue boundary with resolution
of 10 seconds in time and 20 micrometer in position. A suitable microscope
is used to observe the microelectrodes and the tissue near which they are
moved.
In the diagram a microelectrode, whose tip is filled with the liquid
ion exchanger LIX, is initially at a distance x from the tissue into which
ions are moving with a net flux J. 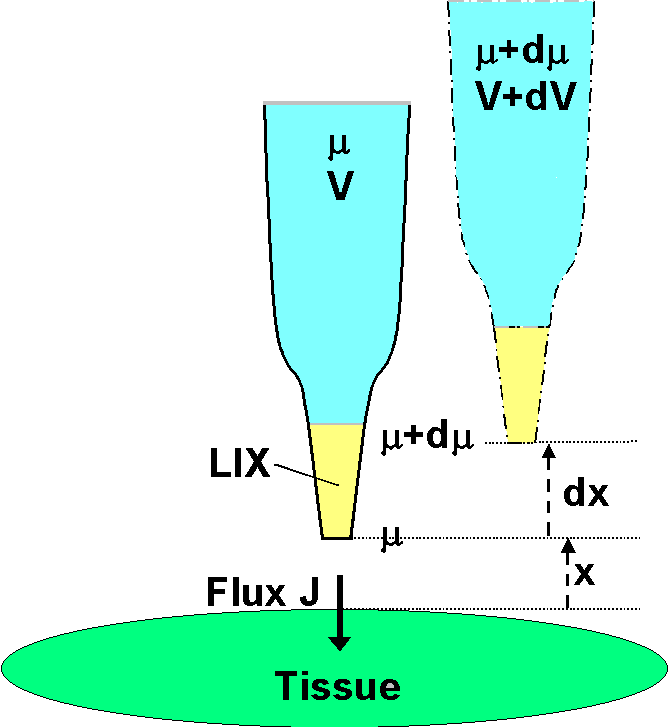 It
is assumed that there is no bulk solution flow, so that ionic movement
in the solution (regardless of the membrane transport processes) is solely
by diffusion under the influence of electric and chemical forces in solution.
It is also assumed that the measurement is close to the surface that the
ionic movement is normal to the surface. The electrochemical potential
in the solution at the distance x is µ (joules mol-1).
Because the LIX allows free passage of the ion in question (but no others),
the electrochemical potential of the ion inside the electrode is also µ.
The chemical component of µ inside the electrode is fixed by the
concentration of the filling solution; the electrical component is given
by zFV , where V (volts) is measured by an electrometer connected
via suitable half cells to the electrode solution and to a reference electrode
some distance away in the bath solution. The ion's valence is
z
and F is the Faraday number. The microelectrode is now moved slowly
(not to disturb the solution) away through a small distance
dx.
(It is shown offset in the diagram for clarity only.) At this new position
in solution the electrochemical potential is µ + dµ.
It is the same inside the electrode, but only the electrical component
there has changed, and the measured voltage is now V + dV.
It
is assumed that there is no bulk solution flow, so that ionic movement
in the solution (regardless of the membrane transport processes) is solely
by diffusion under the influence of electric and chemical forces in solution.
It is also assumed that the measurement is close to the surface that the
ionic movement is normal to the surface. The electrochemical potential
in the solution at the distance x is µ (joules mol-1).
Because the LIX allows free passage of the ion in question (but no others),
the electrochemical potential of the ion inside the electrode is also µ.
The chemical component of µ inside the electrode is fixed by the
concentration of the filling solution; the electrical component is given
by zFV , where V (volts) is measured by an electrometer connected
via suitable half cells to the electrode solution and to a reference electrode
some distance away in the bath solution. The ion's valence is
z
and F is the Faraday number. The microelectrode is now moved slowly
(not to disturb the solution) away through a small distance
dx.
(It is shown offset in the diagram for clarity only.) At this new position
in solution the electrochemical potential is µ + dµ.
It is the same inside the electrode, but only the electrical component
there has changed, and the measured voltage is now V + dV.
Basic electrochemical theory shows that the net ionic flux J
is given in terms of the ion concentration c (mol m-3),
the mobility of the ion u (speed per unit force, m s-1
per newton mol-1), and the force per mole which is the electrochemical
potential gradient (dµ/dx). Thus J = c u
(dµ/dx). But dµ is the same inside the
electrode as in the bath solution, and in the electrode dµ
= zFdV because the concentration inside is fixed. Hence the flux
may be written J = c u z F(dV/dx). The concentration
is known, or is adequately measured by the value of V when the electrode
has been calibrated in standard solutions. For the ion, u and z
are known constants, although for multi-valent
ions u depends on z. The electrometer measures dV as the electrode
is moved through the chosen distance dx.
The theory can also be expressed in terms of the the diffusion coefficient
D
for the ion instead of the (related) mobility u. The theory must
also be modified to apply to spherical or cylindrical tissues. These alternatives,
and the many practical qualifications and limitations, are discussed in
the literature, and particularly in the definitive
review in the January 2001 Plant, Cell & Environment.
There appear to be two systems designed to implement this technique.
Both systems have a sampling rate > 10 Hz and both minimise noise by digital
averaging over longer time periods. Thus it would appear that both approaches
have the same ultimate limitation on their sensitivity which is set by
the thermal electronic noise in the high resistance of the ion selective
liquid ion exchanger (LIX) in the micropipette tip. This theoretical limit
is discussed in Ryan et al. (1990). That and other references
to work using the technique are available.
The MIFE system described below, and in more detail
in the review, was developed
in Tasmania. The other, which is based on the Vibrating Probe, was developed
at Woods Hole in the USA and has been used by Jaffe and various co-workers
(eg Kuhtreiber & Jaffe,
1990). [to top of page]
The MIFETM System
for ion flux measurement
 The
MIFE system uses a stepper motor-driven micromanipulator to move four ion
selective microelectrodes that measure the electrochemical potential of
the ions at two positions in solution close to a tissue surface. Custom-built
electronic amplifiers are provided. The CHART
program is used to control the data acquisition. From the electrochemical
potential difference so measured, the net flux of the ion in solution is
calculated using the MIFEFLUX program.
The
MIFE system uses a stepper motor-driven micromanipulator to move four ion
selective microelectrodes that measure the electrochemical potential of
the ions at two positions in solution close to a tissue surface. Custom-built
electronic amplifiers are provided. The CHART
program is used to control the data acquisition. From the electrochemical
potential difference so measured, the net flux of the ion in solution is
calculated using the MIFEFLUX program.
The MIFE system components developed in Tasmania are available for purchase
on a commercial basis. These are indicated in bold in the following
summary.
System component summary
Electrophysiology lab with bench and Faraday cage.
Microelectrode preparation and filling facilities.
Microscope with vertical or horizontal optic axis as required for the
application.
Hydraulic micromanipulator or piezoelectric translator with its amplifier
and power supply.
Stepper motors and their drives and power supplies (for the hydraulic
manipulator).
Multiple electrode mount.
Manual micromanipulator for adjusting electrode or chamber position.
Computer: PC 286 at least, with DOS, at least 250MB HDD and archiving
facility. DAS08 data acquisition card. MIFE electronics: main amplifier
and two 4-channel preamplifiers.
CHART/MIFEFLUX software.
General purpose software including spreadsheet and (if needed) Borland
Pascal.
Many other things are needed for setting
up a MIFE laboratory to measure ion fluxes. We have provided an outline
of these requirements with estimates of costs and sources of supply.
The CHART program to record data
CHART is the software package, running under DOS, designed to control data
acquisition by the MIFE hardware system. This software allows automated
and interactive real-time control of the amplifier configuration and the
micromanipulator while the data is being collected and written to disk.
The system configuration is recorded together with the data, and all modifications
during data acquisition are recorded in a log file which can also include
annotations typed during the experiment. Up to 8 channels of data are displayed
on the screen as if on a chart recorder. Any region of the chart can
be inspected (and expanded or contracted) at will without interrupting
the measurements. The software allows subsequent recall and display of
any run. It also allows the export of the raw or averaged data in the form
of an ASCII file for import into a spreadsheet or other program. The system
will also function as an excellent electrometer/recorder with a 10 Hz bandwidth
for microelectrode studies of membrane potential or for any other data
acquisition. [to top of page]
The MIFEFLUX program to calculate ion fluxes
MIFEFLUX was developed to implement the flux calculations according to
the published procedures and to provide the necessary software for purchasers
of the MIFE amplifiers and controller. It takes output
files from CHART and produces convenient ASCII text
files for spreadsheet importing. Users who wish to modify the analysis
routines will require their own copy of Turbo Pascal or Borland Pascal
to edit and compile the Pascal source code which will be provided to MIFE
system purchasers. Commercial data manipulating and display software will
be needed to graph the calculated fluxes or other data.
© 1999-2001 University of Tasmania. Back
to main MIFE page. Back to Biophysics home
page. Go to top of this page.
 It
is assumed that there is no bulk solution flow, so that ionic movement
in the solution (regardless of the membrane transport processes) is solely
by diffusion under the influence of electric and chemical forces in solution.
It is also assumed that the measurement is close to the surface that the
ionic movement is normal to the surface. The electrochemical potential
in the solution at the distance x is µ (joules mol-1).
Because the LIX allows free passage of the ion in question (but no others),
the electrochemical potential of the ion inside the electrode is also µ.
The chemical component of µ inside the electrode is fixed by the
concentration of the filling solution; the electrical component is given
by zFV , where V (volts) is measured by an electrometer connected
via suitable half cells to the electrode solution and to a reference electrode
some distance away in the bath solution. The ion's valence is
z
and F is the Faraday number. The microelectrode is now moved slowly
(not to disturb the solution) away through a small distance
dx.
(It is shown offset in the diagram for clarity only.) At this new position
in solution the electrochemical potential is µ + dµ.
It is the same inside the electrode, but only the electrical component
there has changed, and the measured voltage is now V + dV.
It
is assumed that there is no bulk solution flow, so that ionic movement
in the solution (regardless of the membrane transport processes) is solely
by diffusion under the influence of electric and chemical forces in solution.
It is also assumed that the measurement is close to the surface that the
ionic movement is normal to the surface. The electrochemical potential
in the solution at the distance x is µ (joules mol-1).
Because the LIX allows free passage of the ion in question (but no others),
the electrochemical potential of the ion inside the electrode is also µ.
The chemical component of µ inside the electrode is fixed by the
concentration of the filling solution; the electrical component is given
by zFV , where V (volts) is measured by an electrometer connected
via suitable half cells to the electrode solution and to a reference electrode
some distance away in the bath solution. The ion's valence is
z
and F is the Faraday number. The microelectrode is now moved slowly
(not to disturb the solution) away through a small distance
dx.
(It is shown offset in the diagram for clarity only.) At this new position
in solution the electrochemical potential is µ + dµ.
It is the same inside the electrode, but only the electrical component
there has changed, and the measured voltage is now V + dV.