
The MIFETM
system
for non-invasive measurement of specific fluxes in solution
near living
plant or animal tissue

 |
The MIFETM
system
|  |
|
Home Biophysics Lab Shabala Lab MIFE Research Facility MIFE user group Membrane
Transport & Fluxes |
Osmoregulation Other MIFE applications: in Plant physiology, they include stress; adaptation; mineral
nutrition; photosynthesis; long-distance transport; growth & development; water relations;
osmoregulation; hormonal physiology; stomatal physiology; plant movements. Hyperosmotic stress is known to significantly enhance net uptake of inorganic ions into plant cells. Until recent, direct evidence for cell turgor recovery via such a mechanism was still lacking. By combining MIFE ion flux measurements with measuring cell turgor changes (with the pressure-probe technique), direct evidence that inorganic ion uptake regulates turgor in osmotically-stressed Arabidopsis thaliana epidermal root cells were obtained (Shabala and Lew 2002). Immediately after onset of hyperosmotic stress the cell turgor dropped from 0.65 MPa to about 0.25 MPa. Turgor recovery started within 2 to 10 min after the treatment and was accompanied by a significant (30 to 80 nmol m-2 s-1) increase in uptake of K+, Cl- and Na+ by root cells. In most cells, almost complete (>90% of initial values) recovery of the cell turgor was observed within 40 to 50 min after stress onset. Similar results were obtained for other plant tissues (Shabala et al 2000; Chen et al 2007), as well as bacterial (Shabala et al. 2009) and fungal (Lew et al 2006) systems. 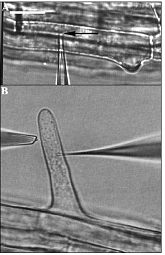 Microphotgraphic examples of pressure probe (A)
and ion-flux/voltage-clamp measurements (B). The micropipette tip is indicated
by an arrow in A. It is located in the vacuole. In B, the double-barrelled
microelectrode is impaled into the cytoplasm. The ion fluxes from the root hair
were measured parallel to the root surface. Bar = 20mm. Microphotgraphic examples of pressure probe (A)
and ion-flux/voltage-clamp measurements (B). The micropipette tip is indicated
by an arrow in A. It is located in the vacuole. In B, the double-barrelled
microelectrode is impaled into the cytoplasm. The ion fluxes from the root hair
were measured parallel to the root surface. Bar = 20mm.
A model illustrating pathways of fast turgor adjustment in Arabidopsis root cells. Hyperosmotic shock, sensed via an osmosensor activates the H+-ATPase. The hyperpolarization increases net K+ uptake thorough an inward K+ channel and concomitantly decreases K+ efflux through an outward K_ channel. Both the hyperpolarized potential and extracellular acidification increase uptake of Cl- through a H+ /Cl- symporter.
A typical recording of the turgor recovery in Arabidopsis epidermal root cell upon onset of hyperosmotic stress.
For more details and reprints, see #9, #12, #13, #14, #28, #39, #55 {{ Needs updatiing }}
Next
|
Maintained by Ian Newman. Date . © University of Tasmania.